A Lithuanian medical company has leveraged Nikon Metrology inspection equipment to advance the production of jaw implants. Ortho Baltic is leveraging AM and metrology technology to make patient-specific implants more affordable and accessible.
Ortho Baltic, based in Kaunas, Lithuania, is one of only three manufacturers of patient-specific temporomandibular joint endoprostheses in Europe. It is also the only one in the Baltic States. In 2012, Ortho Baltic made an investment into additive manufacturing technology, and more recently the company acquired computed tomography (CT) inspection equipment, used for quality control, from Nikon Metrology.
The temporomandibular joint (TMJ) is one of the most used joints in the human body; it allows humans to talk and chew. When this joint is damaged, whether by trauma or illness, and typical treatment methods have proved ineffective, replacement surgery is required. Patient-specific implants are usually reserved for more severe cases, but Ortho Baltic wants to make the administering of tailored endoprostheses the norm.
The company stresses this objective in its mission statement. Ortho Baltic wants to do away with the ‘one implant fits all’ mind-set by making patient-specific implants affordable and accessible.
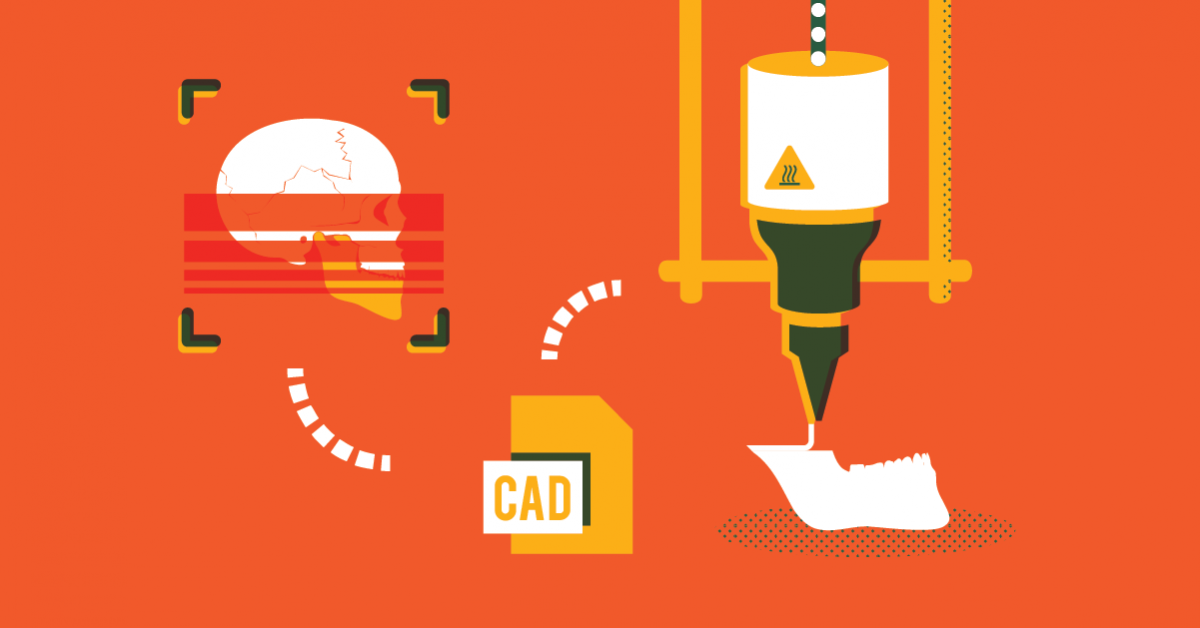
Before production can begin, a series of scans have to take place in order to retrieve the patient’s geometries. From here, implants can be designed with CAD software, and 3D printed. Meanwhile, 3D printing is also used to create surgical guides, which advance the pre-surgical planning.
“To create anatomical models, 3D reconstruction engineers use the patient’s radiological data to perform a 3D reconstruction,” explained Milda Jokymaityte, clinical engineer at Ortho Baltic. “The anatomical bone structures are segmented and virtual 3D models prepared. Including printing, quality control, packaging and sterilisation, the usual lead-time for a patient-specific jaw joint implant is approximately four weeks following the surgeon’s approval of the final design. From this point onwards, pre-surgical planning commences between patient and surgeon, ultimately leading to surgery completion.”
Source: tctmagazine.com